
Students should wash their hands thoroughly before leaving the lab.Always wear safety goggles when handling chemicals in the lab.Small samples of each metal (copper, magnesium, iron, and zinc).750 mL of one of the following solutions: 1.0 M KNO₃, 1.0 M MgSO 4, 1.0 M FeSO 4, 1.0 M CuSO 4, 1.0 M AgNO 3, 1.0 M ZnSO 4.Lesson: 60 minutes (additional time outside of class also needed) Materials (Per lab table) This lab supports students’ understanding of Predict whether or not a chemical reaction will occur.Determine which metals are reactive and which are not as reactive.Grade Levelīy the end of this lab, students should be able to You can design your own simulations.In this lab, students will create an activity series of metals and predict whether or not single replacement reactions are likely to occur. ChemLab – Interactive fee-based service.Late Nite Labs – Includes both Chemistry and Biology labs. 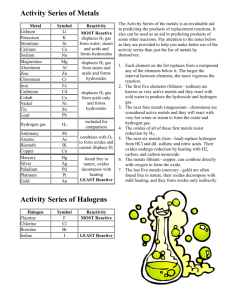
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
